| Model: | - |
|---|---|
| Brand: | HEALGEN;ORIENT GENE;DS |
| Origin: | Made In China |
| Category: | Chemicals / Biochemical |
| Label: | HIV test , home hiv test , rapid hiv test |
| Price: |
-
|
| Min. Order: | 10000 pc |
Product Description
Minimum Order Quantity: 10,000 Piece/Pieces
Supply Ability: 500,000 Piece/Pieces per Week
Payment Terms: L/C,T/T
Packaging & Delivery
Packaging Details: 25 tests/Box or as request
Delivery Detail: 3 ~ 4 weeks
Specifications
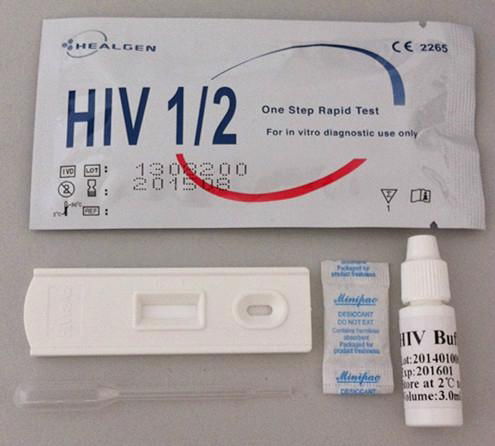
HIV 1/2 One Step Rapid Test Cassette
CE Certificate (CE 2265)
ISO 13485 Certificate
HIV 1/2 One Step Rapid Test Cassette
Intruduction:
HIV is the etiologic agent of Acquired Immune Deficiency Syndrome (AIDS). Of all the HIV types, HIV type 1 has been isolated from patients with AIDS and AIDS-related complex, and from healthy people with a high risk for developing AIDS and HIV type 2 has been isolated from West African AIDS patients and from seropositive asymptomatic individuals. Both HIV-1 and -2 elicit immune responses in human. Testing human serum or plasma for the presence of HIV antibodies has proven an effective aid in determining whether an individual has been exposed to HIV and in screening blood products for possible HIV contamination.
Principle:
The HIV 1/2 One Step Rapid Test Cassette is a lateral flow chromatographic immunoassay based on the principle of the double antigen–sandwich technique. The membrane is coated with recombinant HIV antigens on the test line region of the device. When a specimen is applied at one end of the membrane, it reacts with HIV antigen coated gold conjugate in the test. The mixture then migrates chromatographically by capillary action and reacts with the recombinant HIV antigens on the membrane in the test line region.
If the serum or plasma contains antibodies to HIV-1 or HIV-2, a coloured line will appear in the test line region, showing a positive result. The absence of the coloured test line indicates that the serum or plasma does not contain the anti-HIV antibodies, showing a negative result. A coloured line will always appear at the control line region to serve as a procedural control.
This indicates if the proper volume of specimen has been added and that membrane wicking has occurred.
Materials Supplied:
Each test contains :
1. One cassette device
2. One pipette dropper
3. One desiccant
Each kit contains:
1. 25 test devices
2. One instruction
3. One bottle of Buffer
HIV 1/2 Rapid Test Strip
Minimum Order Quantity: 20,000 Piece/Pieces
Supply Ability: 500,000 Piece/Pieces per Week
Payment Terms: L/C,T/T
Packaging & Delivery
Packaging Details: 100 tests/Box or as request
Delivery Detail: 3 ~ 4 weeks
Specifications
HIV 1/2 Rapid Test strip
CE Certificate (CE 2265)
ISO 13485 Certificate
Intruduction:
HIV is the etiologic agent of Acquired Immune Deficiency Syndrome (AIDS). Of all the HIV types, HIV type 1 has been isolated from patients with AIDS and AIDS-related complex, and from healthy people with a high risk for developing AIDS and HIV type 2 has been isolated from West African AIDS patients and from seropositive asymptomatic individuals. Both HIV-1 and -2 elicit immune responses in human. Testing human serum or plasma for the presence of HIV antibodies has proven an effective aid in determining whether an individual has been exposed to HIV and in screening blood products for possible HIV contamination.
Principle:
The HIV 1/2 Rapid Test Strip is a lateral flow chromatographic immunoassay based on the principle of the double antigen–sandwich technique. The membrane is coated with recombinant HIV antigens on the test line region of the device. When a specimen is applied at one end of the membrane, it reacts with HIV antigen coated gold conjugate in the test. The mixture then migrates chromatographically by capillary action and reacts with the recombinant HIV antigens on the membrane in the test line region.
If the serum or plasma contains antibodies to HIV-1 or HIV-2, a coloured line will appear in the test line region, showing a positive result. The absence of the coloured test line indicates that the serum or plasma does not contain the anti-HIV antibodies, showing a negative result. A coloured line will always appear at the control line region to serve as a procedural control.
This indicates if the proper volume of specimen has been added and that membrane wicking has occurred.
Member Information
| Zhejiang Orient Gene Biotech Co., Ltd | |
|---|---|
| Country/Region: | Zhe Jiang - China |
| Business Nature: | Manufacturer |
| Phone: | 71972583 |
| Contact: | Caroline (Sales Manager) |
| Last Online: | 19 Nov, 2014 |